Name of Project: Crazy Crystals
Question: Does temperature affect the growth of crystals?
Hypothesis:If the solution is kept cold it will grow better and bigger crystals.
Variables:
Independent:Changing the temperature.
Controlled:The materials that i will be keeping the same are pencils, solution, and jars.
Dependent:Observing what happens with the crystals inside the jar.
Materials and Tools:
1. In this science fair project we will be recrystallizing borax under three different temperature conditions: in the refrigerator, at room temperature, and in ice bath.
2. Prepare an ice bath by filling the large bowl half full of ice and then add water three-quarters full.
A. Place the ice bath on a counter top or table, were it can be left undisturbed for at least three days.
3. As soon as the ice bath is prepared, use the thermometer to take the temperature of the ice bath put the thermometer in the bowl were you put the ice and water. Record the temperatures in your lab notebook.
4. Cut three pieces of string and tie one around each pencil. The string pieces should be of equal length and should be long enough that when the pencil is laid across the top of the jar, the end of the string hangs down to just above the bottom of the jar.
5. Bring enough water to fill each jar three-fourths full to a boil in a pot, with adult supervision.
6. Add 1 tablespoon (Tbsp.) of borax to the water, and stir until it dissolves. Repeat, 1 Tbsp. at a time, until no more borax will dissolve. This is your saturated solution.
7. With an adult's help, pour equal amounts of the saturated solution into the three jars. The jars should be about three-fourths full.
8. Lay a pencil across the top of each jar so the strings hang down into the saturated solution.
9. Cover the jars with plastic wrap, wax paper, or aluminum foil.
10. Place one jar in the refrigerator, leave one undisturbed on a countertop or table at room temperature, and put one in the ice bath you prepared.
11. Leave the jars alone for a minimum of 3 days or until crystals form (whichever is longer), and be sure not to disturb them. Check the ice bath regularly to make sure that the ice has not melted. Add ice as necessary.
A. If crystals form under one condition before they do in the others, note that in your lab notebook and let all three conditions continue for another hour to see if any crystals form in the other conditions.
B. Record in your lab notebook the total amount of time (from step 9 to step 11) that you let the crystals form.
12. Carefully remove the pencils, one at a time, and note the size, shape, and number of crystals obtained from each solution. Are there any differences? Why do you think this is so? Record your observations in a data table, like the one below.
Research:
Crystals are structures of regular repeated patterns from connected atoms. Crystals grow from a process called nucleation. In a nucleation molecules will crystallize & dissolve into groups in a solvent. Solute particles contact each other and become bigger. This particle is bigger than a particle by itself. So more particles will connect with that particle and connect with it.(This crystal nucleus becomes bigger,then it falls out.)Solute molecules will continue to connect to the surface of the crystal,making it grow until balance is reached between the solute molecules in the crystal and the ones that are still in the solution.
In order to grow a crystal you need to make a solution. To make a chance for the molecules to come together in a group and form a nucleus.(It also grows in your crystal so in order for it to grow,you will want a concentrated solution with as much solute that you can dissolve.)
A rough surface tends to be more attractive for nucleation then a smooth surface. For example a crystal is more likely to start forming on a rough piece of string, than on a smooth side of glass
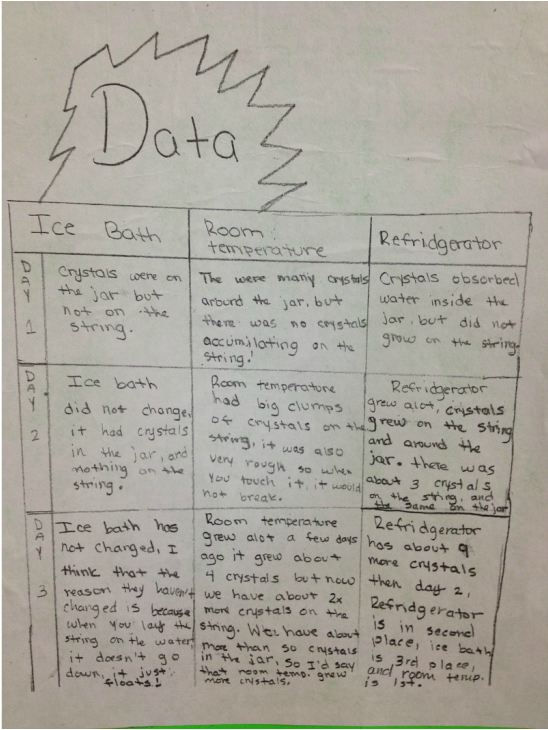
Results:See Below:)
Conclusion:Our hypothesis was wrong because crystals did not grow best in the cold temperature. Our data shows that crystals did not grow at all in the ice bath which is the coldest temperature. Instead of the cold temperature growing more crystals the room temperature had the most crystals.
Hypothesis:If the solution is kept cold it will grow better and bigger crystals.
Variables:
Independent:Changing the temperature.
Controlled:The materials that i will be keeping the same are pencils, solution, and jars.
Dependent:Observing what happens with the crystals inside the jar.
Materials and Tools:
- Lab notebook
- Large bowl
- Ice, enough to fill large bowl at least three times
- Water
- Thermometer
- String
- Scissors
- Pencils (3)
- Identical jars or large drinking glasses (3)
- Pot with a lid
- Borax
- Tablespoon
- Plastic wrap, wax paper, or aluminum foil Gloves, latex or similar style exam glove (optional). Can be used if there is concern over handling borax.
1. In this science fair project we will be recrystallizing borax under three different temperature conditions: in the refrigerator, at room temperature, and in ice bath.
2. Prepare an ice bath by filling the large bowl half full of ice and then add water three-quarters full.
A. Place the ice bath on a counter top or table, were it can be left undisturbed for at least three days.
3. As soon as the ice bath is prepared, use the thermometer to take the temperature of the ice bath put the thermometer in the bowl were you put the ice and water. Record the temperatures in your lab notebook.
4. Cut three pieces of string and tie one around each pencil. The string pieces should be of equal length and should be long enough that when the pencil is laid across the top of the jar, the end of the string hangs down to just above the bottom of the jar.
5. Bring enough water to fill each jar three-fourths full to a boil in a pot, with adult supervision.
6. Add 1 tablespoon (Tbsp.) of borax to the water, and stir until it dissolves. Repeat, 1 Tbsp. at a time, until no more borax will dissolve. This is your saturated solution.
7. With an adult's help, pour equal amounts of the saturated solution into the three jars. The jars should be about three-fourths full.
8. Lay a pencil across the top of each jar so the strings hang down into the saturated solution.
9. Cover the jars with plastic wrap, wax paper, or aluminum foil.
10. Place one jar in the refrigerator, leave one undisturbed on a countertop or table at room temperature, and put one in the ice bath you prepared.
11. Leave the jars alone for a minimum of 3 days or until crystals form (whichever is longer), and be sure not to disturb them. Check the ice bath regularly to make sure that the ice has not melted. Add ice as necessary.
A. If crystals form under one condition before they do in the others, note that in your lab notebook and let all three conditions continue for another hour to see if any crystals form in the other conditions.
B. Record in your lab notebook the total amount of time (from step 9 to step 11) that you let the crystals form.
12. Carefully remove the pencils, one at a time, and note the size, shape, and number of crystals obtained from each solution. Are there any differences? Why do you think this is so? Record your observations in a data table, like the one below.
Research:
Crystals are structures of regular repeated patterns from connected atoms. Crystals grow from a process called nucleation. In a nucleation molecules will crystallize & dissolve into groups in a solvent. Solute particles contact each other and become bigger. This particle is bigger than a particle by itself. So more particles will connect with that particle and connect with it.(This crystal nucleus becomes bigger,then it falls out.)Solute molecules will continue to connect to the surface of the crystal,making it grow until balance is reached between the solute molecules in the crystal and the ones that are still in the solution.
In order to grow a crystal you need to make a solution. To make a chance for the molecules to come together in a group and form a nucleus.(It also grows in your crystal so in order for it to grow,you will want a concentrated solution with as much solute that you can dissolve.)
A rough surface tends to be more attractive for nucleation then a smooth surface. For example a crystal is more likely to start forming on a rough piece of string, than on a smooth side of glass
Results:See Below:)
Conclusion:Our hypothesis was wrong because crystals did not grow best in the cold temperature. Our data shows that crystals did not grow at all in the ice bath which is the coldest temperature. Instead of the cold temperature growing more crystals the room temperature had the most crystals.